A Clinical Trial For Patients With
Myelofibrosis
Have you or a loved one been diagnosed with myelofibrosis and considering treatment with an approved JAK inhibitor?
You could be eligible to participate in a clinical trial for patients with myelofibrosis.
About Myelofibrosis
Myelofibrosis is a rare cancer that disrupts the normal production of blood cells in the human body. Myelofibrosis causes the formation of scar tissue or fibrosis in the bone marrow, impairing the bone marrow's ability to produce healthy red blood cells and platelets. The loss of red blood cells and platelets can lead to anemia and thrombocytopenia, which may cause weakness, fatigue and life threatening bleeding events.
As the bone marrow makes fewer and fewer healthy red blood cells, abnormal growth of blood-cell forming cancer cells may occur outside of the bone marrow in organs such as the spleen or liver causing them to become enlarged. An enlarged spleen is common in patients with myelofibrosis, and it may result in abdominal pain/discomfort, a feeling of early fullness and a decreased appetite.
Cancer cells produce substances called cytokines that cause cancer-related inflammation. The abnormal release of these cytokines results in disease related symptoms such as fatigue, night sweats, itching, and fevers.
Patients with myelofibrosis typically need treatment. However, approved therapies don’t benefit all patients. Despite treatment, patients’ spleens may remain enlarged, and symptoms may persist. These patients, who have a suboptimal response to approved therapies, may benefit from the addition of another treatment to help reduce their spleen size and decrease their symptoms further.
Patients with myelofibrosis are typically prescribed a Janus Kinase inhibitor (JAK inhibitor) that reduces the size of an enlarged spleen and relieves the symptoms associated with myelofibrosis-related inflammation. Unfortunately, current approved JAK inhibitor therapies in myelofibrosis may not reduce spleen size nor relieve symptoms enough. JAK inhibitors have also shown limited impact on restoring normal blood production or reversing diseases-associated bone marrow fibrosis.
Navtemadlin
Navtemadlin is an oral investigational therapy that targets a protein called MDM2, offering a potential new treatment for patients with myelofibrosis.
Increased MDM2 levels in myelofibrosis cancer cells decrease the activity of p53, a tumor suppressor protein. p53 plays an important role in the body’s natural defenses against cancer. In myelofibrosis, cancer cells abnormally increase MDM2 levels to prevent p53 from carrying out its usual cancer-fighting functions.
In patients with myelofibrosis, navtemadlin treatment inhibits MDM2 to restore the ability of p53 to kill myelofibrosis cancer cells.
POIESIS Trial
POIESIS is a global phase 3 registration study for patients with myelofibrosis whose disease has a suboptimal response following treatment with the JAK inhibitor, ruxolitinib – that is, their spleens remain enlarged, and symptoms persist. The study will compare the efficacy and safety of adding navtemadlin to ruxolitinib versus adding placebo to ruxolitinib.
JAK inhibitors are used in the first line setting for patients with myelofibrosis and are the only approved treatment options. There are currently no approved therapies for patients who have not optimally responded to treatment with a JAK inhibitor. The hope is that navtemadlin may provide additional benefit over and above shrinking the spleen and reducing myelofibrosis-related symptoms (e.g. reducing bone marrow fibrosis and the number of myelofibrosis cancer cells).
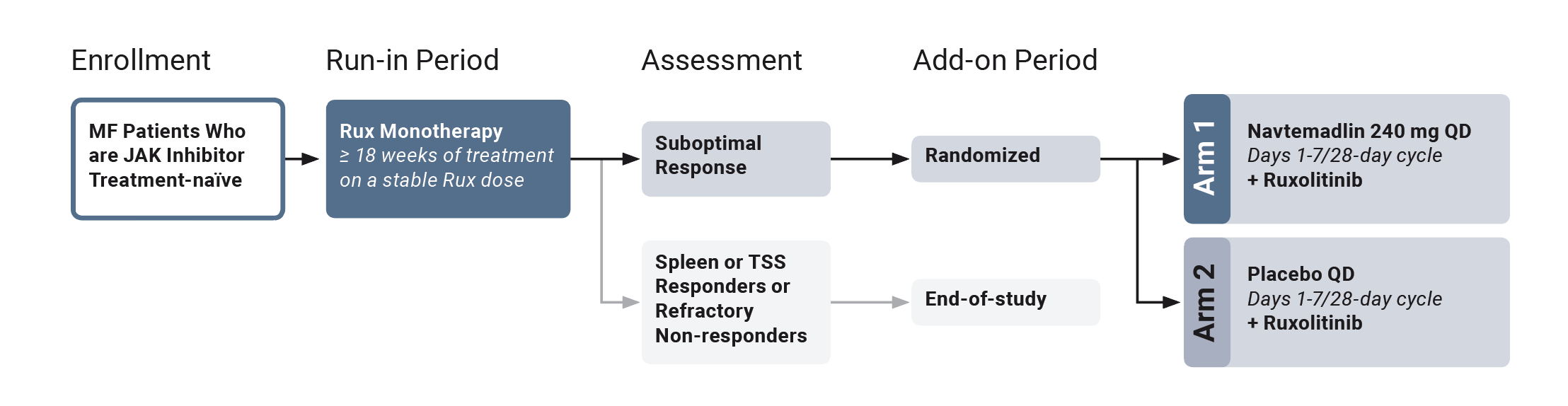
There are two parts to this study: Run-in period and Add-on period.
Part 1: Run-in Period
- Should you meet the eligibility criteria and have never been treated with a JAK inhibitor, you will enter the run-in period and take ruxolitinib for 18-24 weeks. The dose of ruxolitinib will be determined by your study doctor.
- If your ruxolitinib treatment is helping to shrink the spleen and reduce symptoms to some degree but is not sufficiently effective (considered “sub-optimal”) and you meet other study criteria, you will proceed to the second part of this study (randomized add-on period).
- If you respond well (“optimally”), you will not proceed to the second part of this study but may remain on your current ruxolitinib treatment. You will no longer be a participant in the study.
- Should you not respond at all to treatment with ruxolitinib, you will not proceed to the second part of this study. Your doctor will work with you to determine your treatment after you leave the study.
Part 2: Add-on Period
- If you meet criteria for the randomized add-on period, you will be assigned to receive either add-on navtemadlin or add-on placebo, while continuing ruxolitinib treatment at the same dose that was established during the run-in period.
- In the randomized period, 180 patients will be randomized in a 2:1 fashion to receive either navtemadlin (120 patients) or placebo (60 patients). You therefore have a 2 out of 3 chance of receiving navtemadlin.
- This part of the study is blinded, meaning the patients, doctors and the sponsor will not know which add-on treatment (navtemadlin or placebo) is being taken.
Before Treatment Starts
All patients will first undergo complete clinical evaluations. Blood samples will be collected, spleen volume will be measured using imaging scans (such as MRI or CT), and patients will fill out questionnaires to assess the severity of their myelofibrosis-related symptoms. Upon consent of study participation and during the study period, these procedures (including but not limited to blood work, spleen imaging and the daily 7-symptom questionnaire) will be ongoing until week 28 of the randomized period or end of treatment, whichever is later .
For both periods of the study (run-in period and add-on period), patients remain on study unless they cannot tolerate the treatment or their disease progresses.
Efficacy Analysis
24 weeks after the start of the add-on period, efficacy will be analyzed by comparing navtemadlin to placebo therapy using two key measures:
- Spleen volume reduction by MRI or CT imaging
- Symptom score reduction by the self-reported daily 7-symptom questionnaire
It is very important that MRI/CT imaging appointments are attended, and daily symptom scores are accurately recorded until week 28 or end of treatment, whichever is later.
Click to enlarge
FAQ
A clinical trial is a research study conducted in people with the goal of identifying a new drug or treatment or a new procedure or device. The purpose of a clinical trial is to help doctors identify better or safer ways to treat a disease, or ways to improve the quality of life for patients with a disease. Clinical trials in cancer may be used to test new treatments, new ways to diagnose or prevent cancer, or better ways to manage the symptoms of cancer, as well as the side effects of treatment.
For more information about clinical trials, please review the resources provided by the American Cancer Society, National Cancer Institute, and European Federation of Pharmaceutical Industries and Associations.
Clinical trials are important because they help doctors to identify new medicines that are more effective and safer than currently available treatments. Over the years, many successful cancer treatments have been identified through clinical trials. Clinical trials help doctors to gain a better understanding of cancer and also provide more options for cancer treatment in the future.
For more information about clinical trials, please review the resources provided by the American Cancer Society, National Cancer Institute, and European Federation of Pharmaceutical Industries and Associations.
Clinical trials are conducted in a series of steps or phases, with each phase designed to answer specific questions.
Phase 1: These are the first studies of a new drug conducted in people. The main purpose of a phase 1 trial is to find a safe dose for the new treatment and also to decide how the new treatment can be given (by mouth, by injection, etc). Approximately 15-30 patients participate in a phase 1 trial.
Phase 2: A phase 2 trial is conducted to see if the new treatment works in certain types of cancers. Doctors may study whether the cancer shrinks or disappears, how long it takes for the cancer to come back, or whether the new treatment improves quality of life. Between 25 and 100 patients take part in a phase 2 trial.
Phase 3: Before a new treatment receives full regulatory approval, its safety and efficacy must be compared to the current standard treatment in a large study. Patients are randomly assigned (also called randomized) to either the standard treatment or the new treatment. Placebo-controlled studies are a standard method of clinical investigation for new drugs. Phase 3 studies have a placebo (sugar pill) control arm when there is no standard treatment available and patients in the placebo group would not receive any treatment if they were not in the trial. Phase 3 trials typically include several hundred patients.
For more information about clinical trials, please review the resources provided by the American Cancer Society, National Cancer Institute, and European Federation of Pharmaceutical Industries and Associations.
The costs associated with a clinical trial include doctor visits, hospital stays, cost of drugs, lab tests, X-rays and other imaging tests. These costs may be covered by your health insurance or the study sponsor (in this case, Kartos Therapeutics).
Other costs that you may incur, such as for transportation or housing, may be reimbursed. Please contact your participating cancer center for more information.
If you believe you may have experienced an adverse event while participating in a study that includes navtemadlin, please immediately report this to your physician.
Trial Locations
The POIESIS trial will be conducted in approximately 220 cancer centers across multiple countries. Please use the interactive map below to locate a cancer center near you where patients are being enrolled to this trial. 180 participants are expected to take part in the add-on period of the study.
Do You Qualify?
- You are 18 years of age or older
- You have received a diagnosis of primary or secondary myelofibrosis
- You have not already been treated with a JAK inhibitor such as ruxolitinib
- You are not immediately eligible for a bone marrow transplant
Many other conditions must be met before you may participate in the trial. Please contact your nearest participating center for more information about the requirements for the trial.
If you would like to learn more about POIESIS Clinical Trial for Patients with Myelofibrosis, please fill out the form below.